
For use in lighting, a layer of quantum dots can be sandwiched in between two electrically conductive layers. This exceptionally high efficiency make quantum dots appealing for use in lights and as individual colour pixels in vibrant colour flat panel displays. The energy emitted from quantum dots as light, is close to 100% of the energy put into the system. This avoids poisoning healthy cells and therefore the awful side effects associated with cancer treatments. In this manner, quantum dots can be tuned to seek out cancer cells and deliver chemotherapy drugs directly to the cancer cells. This can include various drugs for treating a disease the quantum dot has been tuned to find. This leaves a large amount of room to place other things on the dot. The number of receptors required on the surface of the dot is small compared to the surface area of the dot itself. Due to the fluorescent nature of quantum dots the site of the problem is then made easily visible. The quantum dots will then seek out and attach to the disease en masse. These receptors can correspond to particular diseases, viruses or other items. Quantum dots can be encased within a shell tuned to mimic organic receptors within the body. Medical Applications and Cancer Treatments 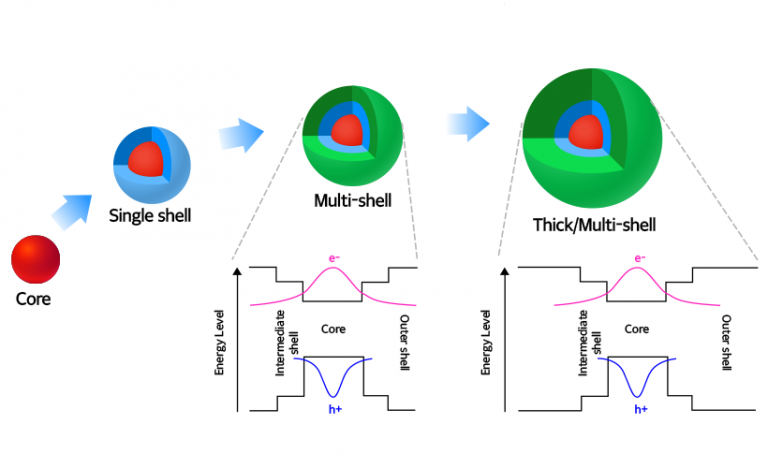
Quantum dots show promise for use in a wide range of applications from the quantum computers of the future, to medical applications, high resolution television screens and household lighting. Electrochemical techniques and CVD can be used to create ordered arrays of quantum dots on a substrate material. The cheapest and simplest method is benchtop colloidal synthesis. Quantum dots can be manufactured by a number of processes from colloidal synthesis to chemical vapour deposition (CVD). By combining a range of sizes of quantum dots in the same sample, the entire light spectrum can be produced simultaneously and appears as white light. In terms of colour in the visible spectrum, this means large quantum dots produce red light and small quantum dots produce blue light – sizes in between account for all the other colours in the spectrum. Large quantum dots produce light with a long wavelength and small quantum dots produce light with small wavelengths. The frequency or colour of light given off is not related to the material used in the quantum dot, but by the size of the quantum dot. The energy source used to stimulate a quantum dot is commonly ultraviolet light. Quantum dots work in much the same way but a quantum dot crystal acts as one very large atom. When the electron returns to it’s lower and stable state, this additional energy is emitted as light corresponding to a particular frequency. When energy is applied to an atom, electrons are energised and move to a higher level. In order to avoid toxic materials leaching from the quantum dots, they are also coating in a protective polymer. Many of the promising applications for quantum dots will see them used within the human body. Quantum dots can be made from a range of materials, currently the most commonly used materials include zinc sulphide, lead sulphide, cadmium selenide and indium phosphide. How many atoms are included in the quantum dot determines their size and the size of the quantum dot determines the colour of light emitted. Quantum dots are tiny nanocrystals that glow when stimulated by an external source such as ultraviolet (UV) light.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
